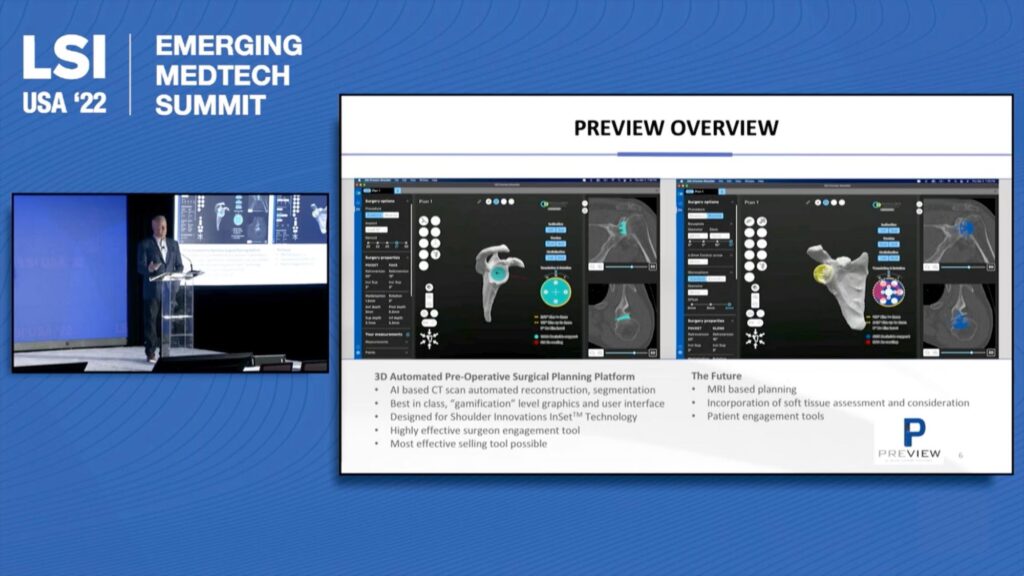
Shoulder Innovations Presentation at 2022 LSI Conference
Recently, David Blue and Matt Ahearn presented on the latest with Shoulder Innovations at the 2022 LSI Emerging Medtech Summit. Watch the replay here:
Shoulder Innovations Presentation at 2022 LSI Conference Read More »